Plastic Bottle Leaching: The Chemical Soup You’re Drinking in Slow Motion
When you pick up a plastic water bottle, it looks clean, transparent, and harmless. It feels like a perfectly sealed container that simply holds water until you drink it. But modern polymer science tells a more complicated story. This happened with all the plastic bottles?
At the molecular level, that bottle is not an inert shell. It is a dynamic chemical environment, constantly interacting with the liquid it contains and with the surrounding environment. Over time, molecules within the plastic can migrate into the water you drink.
In other words, what appears to be a stable bottle may actually behave like a very slow chemical diffusion system—what some scientists metaphorically describe as a chemical soup in slow motion.
Understanding how this happens requires a short journey into polymer chemistry, thermodynamics, and environmental toxicology.

The Ghost in the Bottle: The Molecular Science of Leaching
Most disposable water bottles are made from polyethylene terephthalate (PET), a thermoplastic polymer valued for its clarity, strength, and light weight. PET is widely used in food packaging because it is considered relatively stable and safe under normal conditions.
However, from the perspective of polymer physics, PET is not a perfectly rigid structure. Instead, it consists of long molecular chains arranged in semi-crystalline regions and amorphous regions. These chains vibrate constantly due to thermal motion.
Inside this polymer network exist small molecules such as:
Residual monomers from manufacturing
Catalyst residues
Additives used during production
Degradation byproducts formed over time
These compounds are collectively known as non-intentionally added substances (NIAS). According to Nerín et al. (2013), identifying NIAS in food packaging has become one of the biggest challenges in food safety science because hundreds of different compounds can potentially migrate from packaging materials into food or beverages.
The Mechanism of Molecular Migration
Migration occurs because molecules naturally move from regions of higher concentration to regions of lower concentration, a fundamental principle of thermodynamics known as diffusion.
Inside the plastic, the concentration of certain chemicals is higher than in the water contained in the bottle. Over time, this concentration difference drives molecules to move outward. The process can be described through Fick’s laws of diffusion, which govern how particles move through materials.
Several factors influence the speed of this migration:
Temperature: Higher temperatures increase molecular motion.
Time: The longer water remains in contact with plastic, the greater the leaching.
Surface area: Thin plastic walls allow chemicals to reach the liquid more easily.
The “Time × Temperature” Equation
From a data science perspective, migration can be described as a function:
C = f(t, T)
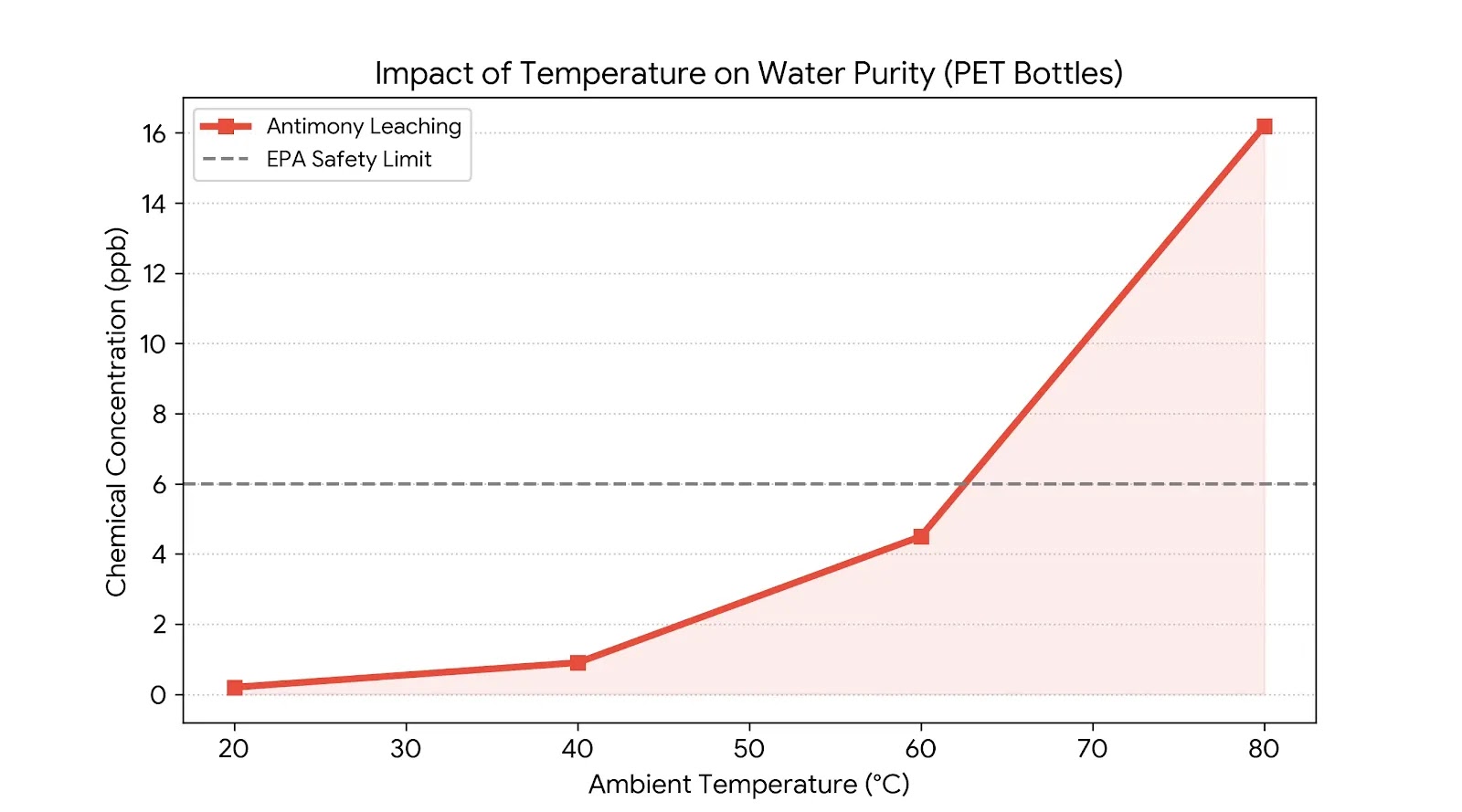
Research published in Water Research by Westerhoff et al. (2008) showed that antimony, a catalyst used in PET manufacturing, can leach into bottled water during storage. Antimony concentrations increased significantly when bottles were exposed to higher temperatures, such as those inside a parked car during summer.
Endocrine Disruptors and the BPA Debate
Another widely discussed issue involves bisphenols. While many bottles are now labeled “BPA-free,” they often use substitutes like Bisphenol S (BPS) or Bisphenol F (BPF). Research suggests that these alternatives may also possess hormone-like biological activity, interacting with estrogen receptors.
Practical Ways to Reduce Exposure
Avoid heat exposure: Do not leave bottled water in hot cars.
Do not reuse disposable bottles: They are not designed for long-term use.
Choose alternative materials: Containers made from stainless steel or glass are chemically stable.
Conclusion: The Data-Driven Choice
What appears to be a simple container is actually a dynamic molecular structure. Switching to a durable reusable bottle, such as an H2GO stainless steel bottle, is not only an environmentally responsible choice but also a practical way to reduce unnecessary chemical exposure.
While chemical leaching is a primary concern, it is only one part of the equation. Recent discoveries show that physical degradation also plays a massive role—specifically through the shedding of nanoplastics in bottled water. Furthermore, even ‘safe’ plastics can be misleading, as we explore in our deep dive into the BPA-free paradox.






