Are Water Bottles Safe? Beyond the Molecule — The Nanoplastics Frontier You Need to Know
Are Water Bottles Safe? Beyond the Molecule — The Nanoplastics Frontier You Need to KnowWhy 240,000 particles per liter should change the way you hydrate.
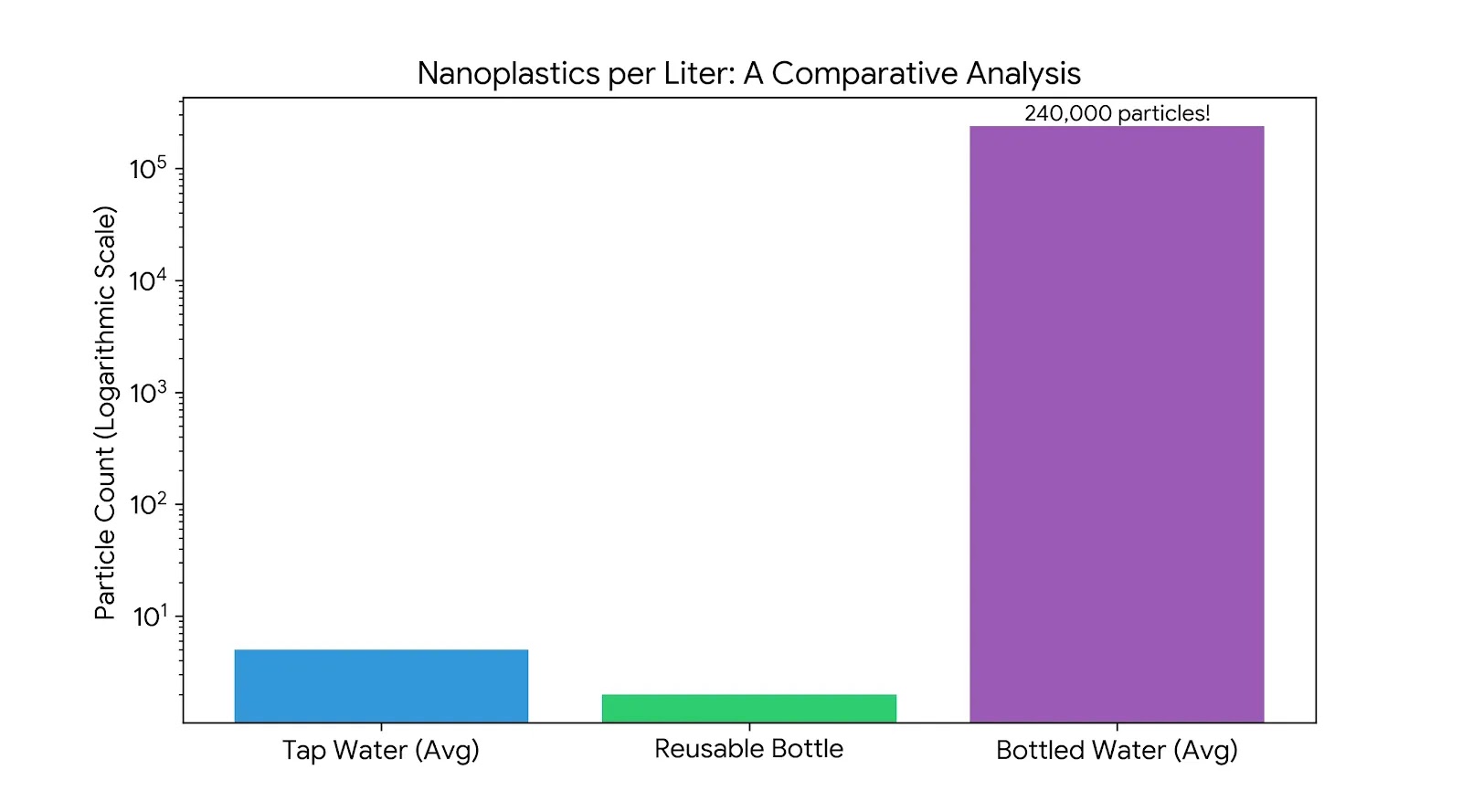
In January 2024, a groundbreaking study published in the Proceedings of the National Academy of Sciences (PNAS) sent shockwaves through the scientific community and global media. For years, we had data on microplastics (particles smaller than 5 millimeters). However, our understanding of the smallest fraction—nanoplastics—remained severely limited by detection technology.
The PNAS study changed everything. Using an advanced hyperspectral Stimulated Raman Scattering (SRS) microscopy platform, researchers analyzed popular brands of bottled water. Their findings were staggering: a single liter of bottled water contains an average of 240,000 detectable nanoplastic particles. so, Nanoplastics in Bottled Water?
This is not just a marginal increase in data; it is a orders-of-magnitude revelation that demands a complete re-evaluation of how we assess the structural integrity of PET packaging.

What are Nanoplastics? Understanding the Scale
While microplastics are small, nanoplastics are truly microscopic. By definition, nanoplastics are plastic particles less than 1 micrometer (1 $\mu$m, or 1000 nanometers) in size.
To understand why 240,000 particles per liter is so alarming, we must understand the biological implications of this scale. The defining characteristic—the “superpower”—of nanoplastics is their ability to breach biological barriers that exclude larger microplastics.
Microplastics (1 $\mu$m – 5 mm): Generally too large to pass through tissue linings; they typically pass through the digestive system unabsorbed.
Nanoplastics (<1 $\mu$m): These are small enough to pass directly through the epithelium of the intestines and the lungs. Once they enter the circulatory system, they become systemic contaminants. Research suggests they are capable of crossing the blood-brain barrier and potentially the placenta.
This microscopic scale moves plastic contamination from a waste management issue to a fundamental metabolic and cellular biology challenge.
The Biological “Trojan Horse”: Surface Area and Toxicity
The danger of nanoplastics is not merely their presence, but their interaction with biological systems, driven by a fundamental principle of material science: the surface-area-to-volume ratio.
As particles get smaller, their surface area relative to their volume increases exponentially. One gram of nanoplastics has thousands of times more active surface area than one gram of large microplastics. This massive relative surface area makes nanoplastics extremely reactive.
This reactivity leads to the “Trojan Horse” effect:
Chemical Adsorption: In the environment or during manufacturing, nanoplastics act as hydrophobic magnets, adsorbing toxic chemicals, heavy metals (like Lead and Mercury), persistent organic pollutants (POPs), and endocrine disruptors (as discussed in Post 2).
Cellular Ingestion: Due to their size, cells confuse these particles for food or natural signaling molecules, ingesting them via endocytosis.
Intracellular Release: Once inside your cells (e.g., in the liver, kidney, or brain), the nanoplastic particle can release this concentrated “cocktail” of adsorbed toxins directly into the cytoplasm or, more critically, into sensitive organelles like the mitochondria.
The Mitochondrial Attack
The most concerning cellular mechanism identified is the potential for nanoplastics to target mitochondria—the powerhouses of the cell responsible for generating ATP (energy).
When nanoplastics (and their adsorbed toxins) infiltrate mitochondria, they disrupt the electron transport chain, leading to the excessive production of Reactive Oxygen Species (ROS). This results in a state known as oxidative stress. Overwhelmed by ROS, mitochondrial membranes are damaged, energy production drops, and the cell may eventually undergo apoptosis (programmed cell death).
This cellular-level breakdown provides a plausible mechanistic link to systemic issues observed in toxicological models, including chronic inflammation and metabolic dysfunction.
Mechanical Shedding: Why Reusable Materials Are Superior
If 240,000 particles are leaching into every liter, we must ask: where is this “plastic dust” coming from? It isn’t just a byproduct of chemical weathering over years; it is actively generated during the normal use of a single-use PET bottle.
The PNAS study and subsequent research identify several primary mechanical sources of particle generation:
Cap Torque and Abrasion: Every time you twist the plastic cap open or closed, the physical friction of the hard polypropylene cap against the PET bottle neck grinds and shears microscopic fragments of plastic directly into the water.
Bottle Compression (Squeezing): Squeezing the flexible bottle during manufacturing, transport, or drinking causes micro-fractures and stress whitening in the polymer matrix, causing surface particles to flake off.
Manufacturing Defects: Standard PET manufacturing (injection stretch blow molding) can leave residual surface imperfections that easily shed particles upon initial filling.
This mechanical vulnerability is the primary engineering difference between single-use PET and high-quality reusable materials like food-grade stainless steel (specifically 18/8 stainless steel, used in H2GO bottles).
A durable stainless steel bottle is designed to be structurally stable. It has high tensile strength and does not suffer from polymer chain fragmentation under mechanical stress or normal thermal fluctuation. While PET packaging is designed to be inexpensive and disposable (prioritizing material lightness over durability), reusable bottles prioritize structural integrity, ensuring that you aren’t drinking “plastic dust” with every gulp.
Securing Your Internal Environment: A Data-Driven Approach
As students of science and advocates of Big Data, we must follow where the evidence leads. The data from 2024 is clear: the volume of systemic plastic contamination we face from bottled water is significantly higher than previously modeled.
While regulatory bodies struggle to update food safety standards to accommodate this emerging data on nanoplastics, we can make informed, data-driven decisions about our own daily routines.
By opting for durable materials like stainless steel and glass, we effectively install a cellular-level “firewall.” We are choosing to secure our internal environment from a pervasive source of structural and chemical disruption. Sometimes, the most technologically advanced solution is simply choosing a better, more stable material.
References
Qian, N., Gao, X., Lang, X., Deng, H., Bratu, T. M., Qiao, Y., Zhang, Y., Lang, G. J., & Min, W. (2024). Rapid single-particle chemical imaging of nanoplastics by SRS microscopy. Proceedings of the National Academy of Sciences, 121(3), e2300582121. [Link to Study]
Mason, S. A., Welch, V. G., & Neratko, J. (2018). Synthetic Polymer Contamination in Bottled Water. Frontiers in Chemistry, 6, 407. [Link to Study]
Vethaak, A. D., & Legler, J. (2021). Microplastics and human health. Science, 371(6530), 672-674. [General toxicology context].






